Manufacturing
thumbnail
Market Trends & Analysis
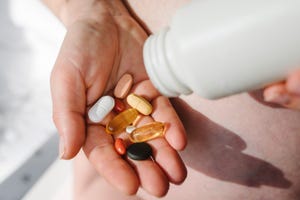
5 reasons capsules are king5 reasons capsules are king
From optimal dosage levels and efficacy to lifestyle preferences, here's why capsules remain the preferred format for 44% of consumers.
Subscribe and receive the latest insights on the health and nutrition industry.
Join 37,000+ members. Yes, it's completely free.






















.png?width=300&auto=webp&quality=80&disable=upscale)






















.png?width=800&auto=webp&quality=80&disable=upscale)