Capitalizing on a targeted, optimized approach to omega-3 supplementation – article
Clinically backed advanced phospholipid omega-3 ingredient, Ruby-O™, provides a new way to optimize EPA and DHA intake
March 3, 2023

Sponsored by Originates
There is no disputing it. Omega-3 fatty acids, especially docosahexaenoic (DHA) and eicosapentanoic (EPA) acids, are critical for the healthful operation of the human body, with a substantial amount of supporting evidence. The trouble is the body cannot produce these essential nutrients on its own and must accumulate them through the diet. Because most American’s don’t eat enough fish containing these healthy fats, the demand for omega-3 fish oil supplements remains important.
In fact, fish/animal oils are among the top-selling ingredients in several key supplement categories, according to data from Nutrition Business Journal’s Condition Specific Report 2022. They are listed as a primary ingredient in categories including support for heart, brain, vision, joint and pre- and post-natal care, and they will represent a nearly $1.3 billion U.S. market in 2023.[1]
But just taking omega-3 supplements is not necessarily enough to meet the body’s needs. Omega-3s are traditionally tough to absorb, so even those taking fish oil may not be getting enough. The market has typically sought ever-higher omega-3 concentrations to address bioavailability and specific health functions. But in this case, more isn’t always better.
Colombia SA-based company Naturmega saw a need for a new strategy in the omega-3 space that not only enhances bioavailability, but also provides a customized, targeted approach to product formulation. Ruby-O™ is an advanced omega-3 phospholipid that represents a more optimized way to formulate with omega-3 fish oils, combining Naturmega’s deep heritage and experience in fish oil manufacturing and product innovation, with added bioavailability and condition-specific concentrations.
First, let’s look at bioavailability. There is ample evidence that phospholipids boost absorption of EPA/DHA.[2] The idea is that phospholipid-based oils disperse better in our water-based cells because of the hydrophilic “head” and the hydrophobic ‘tail”—the whole molecule being key for interaction and absorption. Most of the evidence on absorption centers on krill oil, which contains 40% phospholipids along with EPA/DHA.
Clinical comparison
Naturmega set out to compare Ruby-O’s bioavailability with that of a comparable dose of krill oil in a recent clinical study.[3] The direct comparison was important so that Naturmega could see both the functionality and absorption properties of Ruby-O™, which contains a higher concentration of EPA and DHA, as compared to krill. “The most interesting result we found with the bioavailability study is the concentration and quick rate of EPA and DHA reaching the blood absorption profile,” explained Daniel Minski, director of operation for Originates, the distributor of Ruby-O™ in North America. “The results of this study indicate that the average levels of EPA, DHA and EPA+DHA from a single dose of Ruby-O™ were higher than after a single dose of the krill oil product during the first 10 hours,” he said.
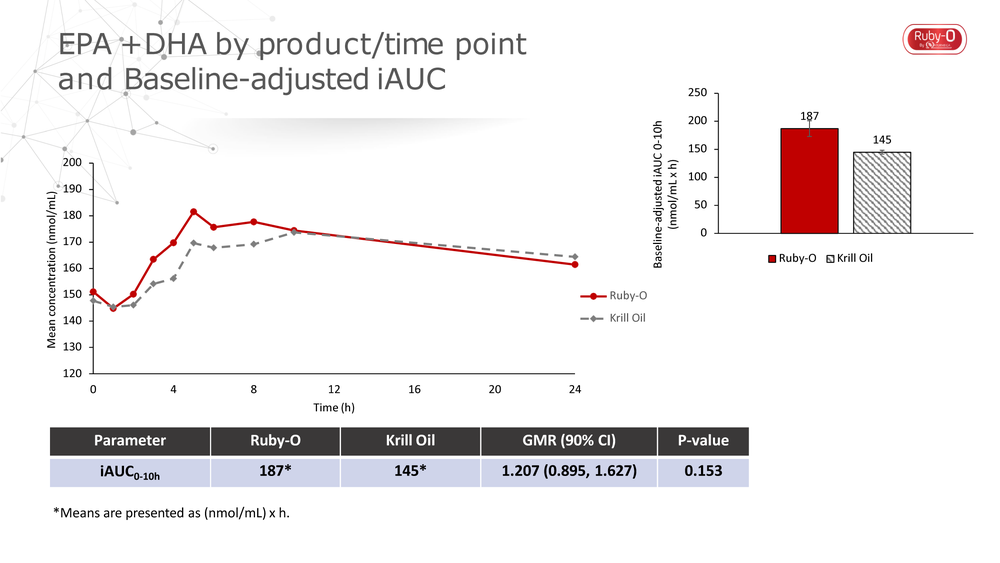
The chart demonstrates how the EPA and DHA concentrations were achieved and faster for Ruby-0™ compared to krill oil. This is attributed to higher and quicker peak values for both EPA and DHA.
The study importantly illustrates both safety and functionality of Ruby-O™, Minski noted. “The data shows that Ruby-O™ provides 64% more EPA+DHA than the krill oil supplement in a single capsule, which resulted in higher AUC for EPA+DHA during the first 10 hours following a single dose.
The findings of EPA+ DHA levels in this study could be compared with other studies in similar conditions, Minski added. With one single dose of 1000mg Ruby-O, the omega-3 levels found were much higher than with an ethyl ester dose with a concentration of EPA + DHA almost 3 times superior.[4]
Customized concentrations
Beyond enhanced bioavailability, Ruby-O™ is also unique because it is produced using high-quality fish oil concentrates and proprietary technology from Naturmega that allows for customized profiles of omega-3 fatty acids. This is crucial, Minski said, because of the deep science on omega-3s. In the European Union, for example, the evidence for omega-3 intake is so convincing that there are nine different approved health claims for EPA/DHA, including recommended maternal intakes for support of a developing fetus and specific intakes for maintenance of normal blood pressure, brain function and vision, with doses ranging from 100mg to 3g.[5]
To create these customized concentrates, Naturmega uses a proprietary technology--molecular distillation--which allows concentration of the EPA and DHA using temperatures below 120°C to avoid deterioration in the oil’s molecular structure.
Of course, the most important raw material in Ruby-O is the fish oil itself, which comes from deep-water fish from the Peruvian coasts considered the cleanest in the world, which means less concern about pollutants. And it�’s suitable for use in a variety of delivery formats including chews, gummies, powders and softgels.
Ultimately, Ruby-O™ is a customizable and sustainable solution for today’s evolving omega-3 market. “Ruby-O´s versatility is important because you can decide the amount of EPA and DHA you need according to your functionality target,” Minski added. “EPA and DHA omega-3s are transported better by Ruby-O´s phospholipids so they can deliver higher and faster EPA and DHA. This capacity of delivering an omega-3 is the one that matters most when thinking in delivery systems, you can use them as a delivery platform.”
[1] Nutrition Business Journal. Supplement Business Report 2022.
[2] Ramprasath VR et al. “Enhanced increase of omega-3 index in healthy individuals, with response to 4-week n-3 fatty acid supplementation from krill oil versus fish oil.” Lipids Health Dis.2013 Dec 5;12:178.
[3] Maki, KC. “A Randomized Double-Blind, Crossover, Kinetic Study to Assess the Relative Bioavailability of Omega-3 Fatty Acids in Two Supplement Products in Health Men and Women.” Midwest Biomedical Research; A Division of MB Clinical Research and Consulting.”
[4] Maki, KC et al. “Effects of a self-micro-emulsifying delivery system formulation versus standard w-3 acid ethyl ester product on the bioavailability of eicosatetraenoic acid and docosahexaenoic acid: a study in healthy men and women in fasted state.” Clin Ther 2018 Dec;40(12):2065-2076.
[5] European Food Safety Authority (EFSA). Scientific Opinion on the Tolerable Upper Intake Level of eicosaepentaenoic acids (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA). EFSA Panel n Dietetic Products, Nutrition and Allergies. EFSA Journal 2012;10(7):2815.
Read more about:
Co branded articlesYou May Also Like




.png?width=800&auto=webp&quality=80&disable=upscale)