Setting the record straight about liposomes
Liposomes are tiny, human-made sacks that encapsulate nutrients, delivering them in a targeted way into the body, but confusion regarding liposomes still exists in the supplements industry.
December 19, 2023
When it comes to essential nutrients like vitamin C, consumers generally understand the benefits. Familiarity is also increasing when it comes to the potential benefits of less popular cutting-edge nutrients like quercetin.
What consumers may not understand is that popping any ol’ quercetin tablet or vitamin C gummy doesn’t always check that benefit box. Different supplements use different delivery systems—and those systems can have a huge impact on how (and whether) the nutrient absorbs.
Enter liposomes.
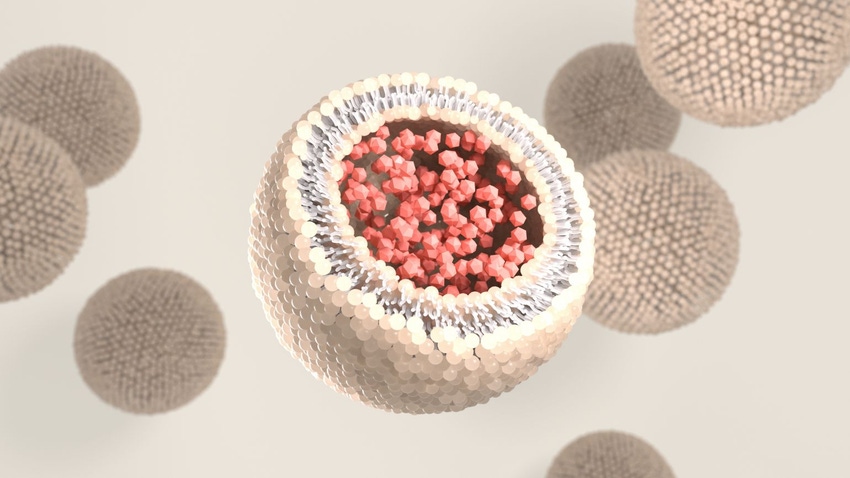
Liposomes are tiny, human-made sacks that encapsulate nutrients, delivering them in a targeted way into the body. They have one or more lipid bilayer membranes (known as phospholipids), making them very similar in structure to human cells. They’re amphiphilic, meaning they have water-soluble and fat-soluble parts; therefore, they can deliver water-soluble or/and fat-soluble nutrients.
“Liposomes really match and mirror our own body's cell structure,” explained Sebastian Balcombe, CEO and founder of ingredient supplier Specnova LLC. “It's how naturally our body likes to absorb things. This isn't something out of left field.”
While this all sounds fairly straightforward, confusion regarding liposomes still exists in the industry. Attempting to set the record straight, Balcombe took part in the “AI, blockchain, regenerative agriculture: Technology’s growing importance in health and nutrition product creation” educational session at this year’s SupplySide West in Las Vegas. His presentation, “Fact & Fallacy in Delivery System Performance,” tackled misconceptions about these innovative delivery systems and dug deeper into their many applications.
Liposome-free liposomal products
“A lot of the products that say they're liposomal, when we test them, they're actually not liposomal at all,” Balcombe pointed out. “Typically, they're simply mixtures of, say, sunflower lecithin as the source of the different phospholipids and the ingredient. That explains why they don't perform better than non-liposomal ingredients.”
Unfortunately, properly validating liposomal ingredients is time-consuming and expensive. A transmission electron cryomicroscopy (CryoTEM) machine—an example of instrumentation needed for validation—can run up to $20 million. And even if a company has access, these unique machines require a specialized skillset. “It’s a whole different world,” Balcombe said.
“I see a lot of these images (from CryoTEMs) and they're pointing to something that's not a liposome,” he continued. “It's actually contamination. With cryo, you're magnifying something 19,000 or 20,000 times, so you're always picking up contamination.”
That’s just one of several possible misinterpretations. “I see images where there's no active ingredient inside,” Balcombe said. “It seems like a lot of the companies don't know how a liposome looks under cryo, let alone some of the other techniques that you need to validate and characterize a liposome.”
With this in mind, it’s important that all liposomal ingredients should have third-party validation. The only problem, according to Balcombe, is there’s no infrastructure for third parties to validate and characterize the liposome. “It’s probably why so many companies can get away with saying, ‘We have a liposome.’ That's a definite weakness.”
The many applications of liposomes
Another fallacy Balcombe dispelled is that liposomes have limited applications.
“We put them in everything from drink powder mixes to capsules to different particle size tablets,” he said. Despite some issues with heat, gummies and chews are also on the table, as the industry continues to work to make liposomes more temperature resistant.
Balcombe noted liposomes can serve several functions and are best known for use in improving solubility and permeability. “These are the ways that people are familiar with liposomes, factors of bioavailability.”
But other factors are also under investigation, such as improving stability. “A lot of natural product ingredients have terrible stability as they go through the body,” Balcombe said. He cited vitamin C as an example, given it is very stable in the low pH of the stomach, but unstable in the higher pH of the small intestines.
Based on an unpublished randomized double-blind placebo-controlled study, Balcombe claims that liposomal vitamin C was 27% more bioavailable in blood plasma compared to non-liposomal vitamin C. Twenty percent more liposomal vitamin C was found in the white blood cells over 24 hours.
The future of liposomal delivery
Research like this vitamin C study is important to the future of liposomes. “We'll keep on pushing on the validation characterization process. We'll keep on building on performance human clinical studies,” Balcombe said.
“At the end of the day, we just want the industry to understand liposomes as much as possible,” he added. “If they truly are liposomes, they're going to perform a lot better than a non-liposomal ingredient. Take an ingredient that already works well and make it a superstar ingredient. That's the dream.”
About the Author(s)
You May Also Like






.png?width=800&auto=webp&quality=80&disable=upscale)