Sports nutrition expert Bruce Kneller reviews the science on SARMs, explaining why the class of substances with many of the desirable effects of testosterone are dangerous.
October 6, 2022

There is much I could write about regarding the ubiquitous availability of SARMs (selective androgen receptor modulators), but I am going to focus on the most critical and misunderstood aspects: Are they (potentially) dangerous and even less safe than anabolic steroids?
Generically speaking, SARMs are a class of androgen receptor agonists that possess many of the desirable effects of testosterone—such as mitigating muscle and bone loss—while possibly decreasing the risks of such undesirable effects as developing prostatic hypertrophy or causing liver damage or cancer. Basically, SARMs strive to produce all the best effects of testosterone while reducing or eliminating the hormone’s potential, undesirable effects.
But I must stress none of the known SARMs fully eliminate the undesirable effects. They may merely reduce some of these to varying degrees.
Most people would refer to SARMs as being “nonsteroidal” in nature. I disagree. Plenty of anabolic steroids that are not testosterone fit nicely into the above definition of a SARM. Among them would be esters of methenolone (Primobolan) and the C-17a methylated anabolic steroid, oxandrolone (Anavar, Oxandrin). Further, I would argue the old anti-breast cancer drug dromostanolone dipropionate (Masteron) fits nicely into the definition of what makes a SARM.
One product being illegally marketed as a SARM—PF-06260414 (aka MK-0773)—is undoubtedly an actual anabolic steroid because it is clearly steroidal in nature. This compound appears widely available online for sale, and in my opinion, is in clear violation of the Designer Anabolic Steroid Control Act of 2014 (DASCA), making its illicit sale in the U.S. not just an FDA issue but quite likely a DEA issue too.
Since I am not a lawyer, I wanted to obtain the thoughts of a legal expert in this arena. I asked my good friend, industry attorney Rick Collins, to opine on the legality of SARMs being sold as both dietary supplements and as research chemicals. Here is what Rick had to say:
“SARMs are mostly sold either as ’dietary supplements’ or as ’research only chemicals not for human consumption.’ They can’t be legally sold as dietary supplements because they don’t meet the legal definition. They can’t be labeled and marketed for ’research only’ if the true purpose is to sell them to fitness enthusiasts for personal physique enhancement. The Food and Drug Administration and the Department of Justice see this ploy as criminal fraud.”
Most people illegally selling and/or using SARMS refer exclusively to those chemicals with a nonsteroidal chemical structure. Therefore, I will limit further commentary to chemicals that are nonsteroidal in nature but may impart some clear, anabolic/androgenic properties in mammalian tissues.
Nonsteroidal SARMs have been in development as Rx drugs by the pharmaceutical industry for at least 25 years. However, FDA has not approved any of these compounds as a human or even veterinary therapeutic drug. Almost all nonsteroidal SARMs are at least loosely, chemically related to an anti-androgen called bicalutamide (Casodex), a potent and prescription drug often used to treat prostate cancer (see Figure 1).
FIGURE 1
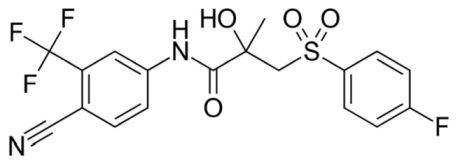
As of 2021, only four nonsteroidal SARMs had gone through bench-top and preclinical development stages required by FDA and have been clinically trialed on humans: MK-2866 (ostarine or enobosarm), LGD-4033 (ligandrol), RAD-140 (testolone) and GSK-2881078.
GSK-2881078 is not yet widely available on the gray or black markets, so I will not discuss this SARM further. Other SARMs either remain in lower phases of development in murine models or have been entirely abandoned for consideration for a multitude of reasons, mostly due to the safety concerns demonstrated in animal studies.
Unfortunately, many of these compounds are readily—albeit illegally—available for purchase under the false pretense they are “dietary ingredients” or “dietary supplements” or are “for sale for research use only.”
SARMs have not been approved by FDA and are associated with serious safety concerns, including potential to increase the risk of heart attack or stroke, and life threatening reactions like liver damage, FDA warned five years ago.
MK-2866 (ostarine or enobosarm)
Ostarine is clearly the most readily available nonsteroidal SARM for illegal purchase. It is ubiquitous and sold openly on the internet and in many “mom & pop” brick and mortar stores. Ostarine’s chemical structure is shown in Figure 2.
Figure 2
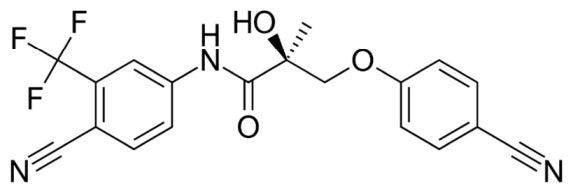
Common adverse side effects associated with the use of ostarine and observed in a clinical study included headache, nausea, fatigue and back pain. Other observed effects were in liver function tests (elevated ALT), reductions in high-density lipoprotein (HDL), blood glucose, insulin and insulin resistance (J Cachexia Sarcopenia Muscle. 2011;2:153-161). A Google search reveals the illicit sale and often suggested use of ostarine on various websites is 10 to 30 times higher than the 1 to 3 mg dose used in the clinical studies.
At least some evidence suggests taking ostarine at these higher doses over just a 12-week time can adversely lead to a marked decrease in endogenous testosterone levels (US Pharm. 2020;45[60]:15-18). Well-known adverse side effects of decreased testosterone include reduced sex drive, erectile dysfunction, infertility, muscle weakness, loss of bone density and weight gain accompanied by increased body fat, insomnia and depression (CMAJ. 2015;187:1369-1377). Ostarine is capable of suppressing endogenous testosterone production, fertility and libido. And these side effects are far more likely to be observed when ostarine is used at 10 to 30 times the dose used in human research studies.
LGD-4033 (ligandrol)
While ligandrol has been “clinically studied” in men, the study (just one Phase II pharmacokinetic heavy study is available publicly) was conducted over 10 years ago and limited to 76 healthy men who received 0.1, 0.3 or 1.0 mg of ligandrol orally on a daily basis for 21 days (J Gerontol A Biol Sci Med Sci. 2013;68[1]:87-95). The study mentions a previous, dose-escalating, Phase I study conducted in humans, but the Phase I study data and analyses have not been publicly released. A Phase II study completed in 2017 that also utilized doses of 0.1, 0.5, or 1.0 mg in older people with hip fractures apparently was also conducted in 108 men and women ages 65 years and older but is unpublished as of now. No further human data is publicly available. Ligandrol’s chemical structure is shown in Figure 3.
Figure 3
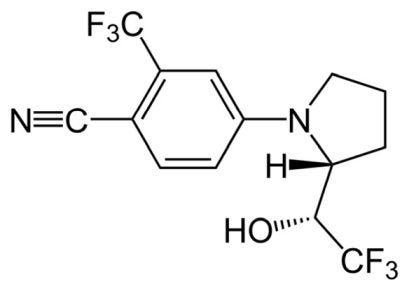
A Google search of the terms “ligandrol” and “bodybuilding” shows “online experts” advocate for the illegal use of this compound at between 5 to 20 mg per day, which is five to 200 times the dosage utilized in research studies in humans. A further Google search of the terms “ligandrol” and “buy” returned mostly solvated, liquid products dispensed via a graduated dropper in concentrations of 10 mg/mL – 30 mg/mL with a few solid-dose products being marketed as containing 5-10 mg per capsule. Ligandrol has been associated with documented and published incidences of significant liver damage. The liver damage was clinically and histologically observable (World J Clin Cases. 2021;9[16]:4062-4071) with as little as two weeks of daily usage.
RAD-140 (testolone)
Much like the SARMs previously discussed, in-human data after exposure to RAD-140 is scant. The first clinical trial was only completed in September 2020. As of today, only a study abstract has been published. This study only enrolled 20 post-menopausal women with breast cancer (median age of subject = 60 years) (Cancer Res. 2020;80[4 Suppl]:P5-11-01). More than 30% of the study subjects experienced elevated liver enzymes (AST & ALT) while on the drug and 80% (16/20) experienced an elevation in PSA. Remember, the study subjects were 100% women!
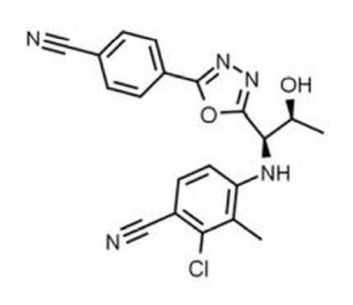
A Google search of the terms “RAD-140” and “buy” demonstrated this SARM is widely available for sale, predominantly in a liquid solution most commonly at 10 mg/mL. Anecdotal feedback shows most users believe between 5 and 30 mg per day is a safe and ideal dose. This suggestion is not based on any known, published clinical data. Testolone’s chemical structure is shown in Figure 4.
Figure 4
It’s what you know for sure that just ain’t so!
Despite the exceedingly limited clinical data regarding the safety and efficacy of SARMs, a proverbial 800-pound pink elephant is standing in front of any would-be purchaser of these products. It’s highly probable what consumers believe they are purchasing and what is being advertised for sale is clearly not what is contained in the product they’re receiving. In a 2017 study of 44 products marketed and sold as SARMs (JAMA. 2017;318[20]:2004-2010), only about half of them (23) contained any SARM at all. Of the 44 tested products, no active compound was detected in four (9%) and substances not listed on the label were contained in 11 (25%). In only 18 of the 44 products (41%), the amount of active compound in the product matched that listed on the label. The amount of compound listed on the label differed substantially from that found by analysis in 26 of 44 products (59%).
Since these compounds are not legal for sale as dietary supplements, the production of any finished product containing them and marketed for human consumption cannot be said to have been produced in a cGMP (current good manufacturing practices)-compliant facility or manner.
A more recent study published tested 111 putative SARM-containing products openly sold online (Perform Enhanc Health. 9[3-4]): “[Q]ualitative chemical analysis revealed that while 94 of the 111 products correctly identified the main active pharmaceutical ingredient on the label, 90 were contaminated with other performance enhancing drugs (emphasis added),” the research found. “Validated quantitative chemical methods assessed dosage strength and homogeneity to reveal that only 34 of the 111 were considered of good (emphasis added) quality.”
In summary, the likelihood that a user of any of these illegal products is getting only what’s declared on the label—and in the amounts advertised—has been shown to be poor. These findings, combined with the research on SARMs’ adverse effects, demonstrates these compounds are truly dangerous.
Bruce Kneller is a longtime sports nutrition formulator/developer and currently is a principal (the “K”) in Millstone, NJ-based KLZ Holdings LLC, along with Hector Lopez, M.D., (the “H”) and Tim Ziegenfuss, Ph.D. (the “Z”). KLZ is an early-stage R&D company with two U.S. patents and one commercialized product in the sports nutrition space (3D Pump Breakthrough).
About the Author(s)
You May Also Like






.png?width=800&auto=webp&quality=80&disable=upscale)