Probiotics can be difficult to test for potency due to the live nature of these ingredients, so testing methods are usually specific to each supplier.
April 26, 2019
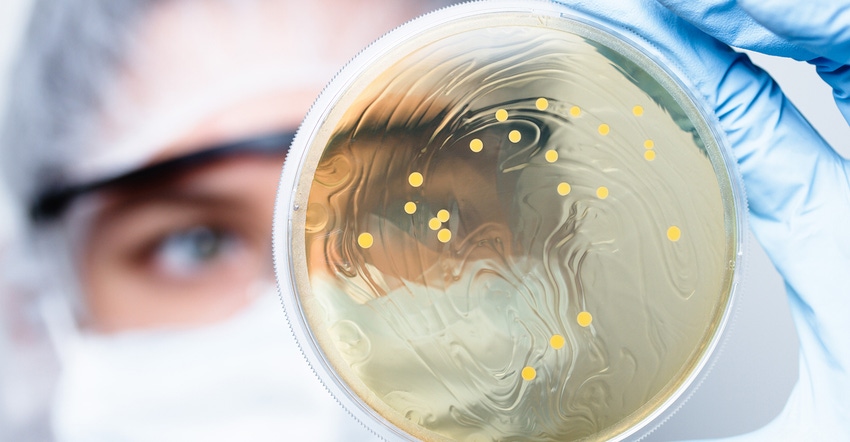
Common digestive health supplement ingredients such as probiotics can be a challenge to understand because these specialty ingredients are unique. For starters, most supplement ingredients are labeled for potency or strength by weight or mg amounts. However, probiotics are labeled using colony forming units (CFU). Weight units may also be listed on a label, but do not reliably confirm that they are active or viable, which is of utmost importance for efficacy. Because of this, testing these specialty ingredients with accuracy is difficult, as the test methods are unusual and may not be run in most labs.
The top goal should always be to manufacture quality products, and following GMPs (good manufacturing practices), along with verification and testing, is necessary to ensure that the identity, purity, strength and composition consistently meets the specification of the product, according to FDA’s Center for Food Safety and Applied Nutrition (CFSAN) “Dietary Supplements—Guidance for Industry: Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements; Small Entity Compliance Guide.”
Probiotic testing methods are usually specific to the supplier and are generally designed for raw material verification. As the probiotic industry grows, more methods are being added to the compendia which compare similar genus probiotic ingredients, but method alignment for the industry is slow. The labeling guidelines, which follow probiotic best practices, as described by the Council for Responsible Nutrition (CRN) and the International Probiotics Association (IPA), are to label single strains or blends with combined strains, in a total CFU/g label claim. Labeling the CFU count for a product containing a single-strain is more straight-forward than enumerating a blended-strain product. The goal is to use one CFU method for obtaining overlapping growth of a multi-probiotic blend. This may take some research and development (R&D) work, in addition to communication with the lab to determine the best method to obtain accurate total CFU/g results.
This article was excerpted from the article “The right testing for probiotic and enzyme applications” from INSIDER’s Digestive Health digital magazine.
Tammy Blakemore is general manager at SORA Labs, a third-party dietary supplement testing lab that specializes in enzyme activity testing and probiotic enumeration.
About the Author(s)
You May Also Like






.png?width=800&auto=webp&quality=80&disable=upscale)