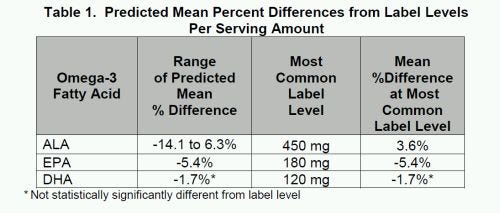
The fatty acid counts on U.S. omega-3 supplement labels meet label claims within 5.5 percent. For the per-serving label amounts, eicosapentaenoic acid (EPA) content was within 5.4 percent of the labeled amounts on average, docosahexaenoic acid (DHA) content was within 1.7 percent, and alpha-linolenic acid (ALA) content was within 3.6 percent.

The fatty acid counts on U.S. omega-3 supplement labels meet label claims within 5.5 percent, according to a new report from the National Institutes of Health (NIH). For the per-serving label amounts, eicosapentaenoic acid (EPA) content was within 5.4 percent of the labeled amounts on average, docosahexaenoic acid (DHA) content was within 1.7 percent, and alpha-linolenic acid (ALA) content was within 3.6 percent.
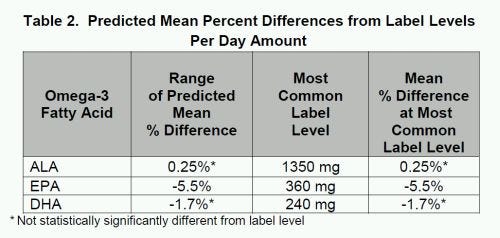
The report also looked at label claims daily omega-3 intakes, as many products suggested multiple servings per day. For the per-day label amounts, the label claims were within 5.5 percent for EPA, 1.7 percent for DHA and 0.3 percent for ALA.
The report, part of the Dietary Supplement Ingredient Database (DSID), examined fish oils (67 of the 89 products in the tests) and flaxseed oils that were purchased in 2010 from retail, Internet and network marketing channels. Researchers compared label claims with omega-3 content of the product based on AOAC analytical methods. However, GOED (the Global Organization for EPA and DHA Omega-3s), USP (United States Pharmacopeia), AOCS (American Oil Chemists' Society) and European Pharmacopeia use other methods for testing omega-3s. Learn more about the differences in omega-3 testing methods in the SupplySide Omega-3 Report “Omega-3 Quality, Testing Methods & Techniques" by Alkemist Labs’ Elan Sudberg.
In a member notification, GOED noted the NIH labelling report does not have practical significance for the market, but it is used to help establish methodologies for estimating actual intakes of nutrients from dietary supplements. For instance, if a consumer is taking a single fish oil softgel that claims to contain 180 mg of EPA, scientists using National Health and Nutrition Examination Survey (NHANES) data will estimate that actual intake was 170 mg based on the results of this study.
Supplements in the study were inconsistent about how they labelled fatty acid content: some products had only a total omega-3 fatty acid content claim, some had only individual omega-3 fatty acid content claims, and some products claimed content levels for both. Approximately 85 percent of the products in the study listed a label amount for at least one of the major omega-3 fatty acids (ALA, EPA and/or DHA).
Thus, Nutrient Data Laboratory (NDL) said in the future, they plan to compare products that labeled individual omega-3 fatty acid content to products that don’t label the ALA, EPA or DHA content.
Also being investigated is how best to define total omega-3 fatty acid levels. Of the 57 fish oil products with a label claim for total omega-3 fatty acids, one-third of the products defined this term by summing EPA and DHA; another one-third defined this term by summing EPA, DHA and other omega-3 fatty acids; and the rest of the products did not define this term. “A possible consensus definition of ‘total omega-3 fatty acids’ is being investigated," the report authors noted.
The DSID also said it was working on developing an online calculators where researchers can enter labeled omega-3 amounts to estimate actual nutrient intakes for research purposes. Currently, it has developed similar calculators for multivitamin and prenatal vitamin supplements.
Additional DSID studies are underway to evaluate ingredient quantities in prescription prenatal multivitamin/minerals (MVMs) and to analyze botanical dietary supplements containing green tea and flavonoids. Estimates of vitamin D, vitamin A and chromium in adult MVMs, previously not reported, will also be released in the future, according to DSID.
The DSID was developed by the Nutrient Data Laboratory (NDL), Beltsville Human Nutrition Research Center, Agricultural Research Service (ARS), USDA and the Office of Dietary Supplements (ODS).
Learn more about the functions and importance of the ODS in the INSIDER Report “Explore and Promote: The Office of Dietary Supplements."
And join Chris Schmidt, Euromonitor International’s consumer health analyst, as he provides a cross-regional overview of omega-3 sales and discusses consumer market trends in the “Trends In Omega-3 Fatty Acids" presentation at SupplySide Marketplace on Wednesday, April 8, 10:30 to 11 a.m., Orlando World Center Marriott.
About the Author(s)
You May Also Like






.png?width=800&auto=webp&quality=80&disable=upscale)